
Greater Long Island coverage is funded in part by Toresco & Simonelli, a boutique injury and family law firm in West Islip. They fight for their clients. Click here to get in touch.
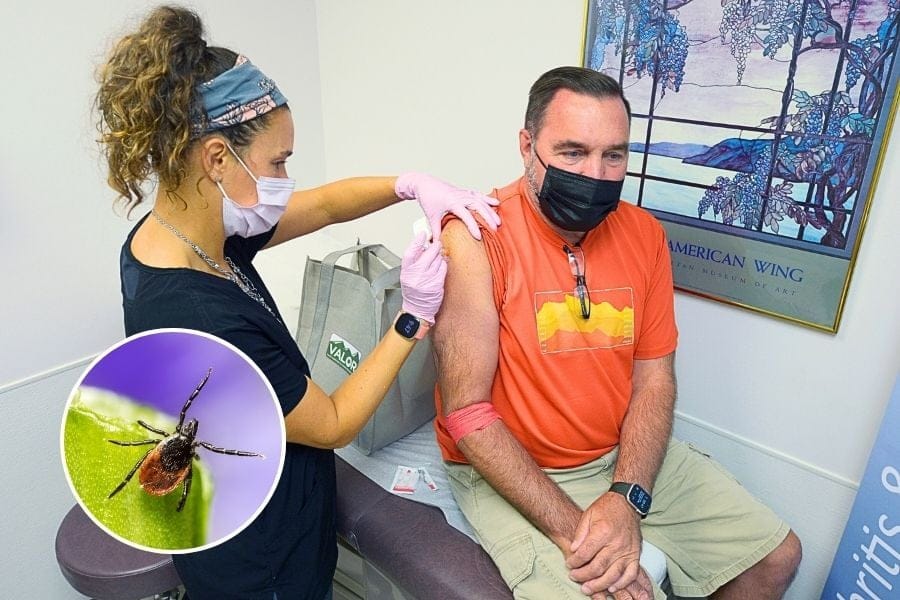
Long Islanders may soon have a shot at avoiding Lyme disease. Quite literally.
A new vaccine developed by Pfizer and French biotech firm Valneva is nearing the end of clinical trials and could be submitted to the FDA as early as 2026, according to Newsday. The companies hope their shot, called VLA15, will succeed where a previous Lyme vaccine fell short.
That earlier vaccine, LYMErix, was introduced in 1998 to much fanfare, but pulled from the market a few years later after complaints of side effects and lawsuits. Though the FDA found no link between the vaccine and any reported injuries, public confidence disappeared and demand dried up.
This time, health experts believe the response could be different.
Lyme disease, once more concentrated on the East End of Long Island, has now spread throughout Suffolk and Nassau counties. Newsday’s report says the Centers for Disease Control and Prevention counted 3,299 cases in Suffolk and 697 in Nassau in 2023 — both increases over the previous year.
Across New York State, more than 21,000 cases were reported, representing nearly a quarter of all cases nationwide. The CDC estimates the real number of cases could be as high as 476,000 annually.
“It’s a problem for everyone across large swaths of land,” Dr. Andrew Handel, a pediatric infectious disease specialist at Stony Brook Children’s Hospital, told Newsday. “I don’t think anyone views Lyme disease that way [as a niche concern] anymore.”
Participants in the VLA15 clinical trial are receiving three doses spaced out over several months, plus a booster shot a year later. The developers say the goal is to build strong, long-term protection — something doctors say is badly needed.
“We see a lot of tick-borne illnesses,” said Dr. Michael Huang, chief of infectious diseases at Peconic Bay Medical Center in Riverhead. “I would tell my patients to consider it because we have a lot of business from Lyme disease, and we don’t need that kind of business.”
Still, Newsday reports that lingering vaccine hesitancy, fueled by misinformation and backlash to pandemic-era mandates, could affect the public’s acceptance of another vaccine. Handel said some families on the East End are eager for the vaccine, while others have flatly rejected the idea.
“Some families desperately want a Lyme disease vaccine so they don’t need to be as worried about tick bites,” Handel told Newsday. “Other families have no interest, saying they would never want their child vaccinated.”
John Gilmore of Long Beach, who heads the Autism Action Network, told Newsday he has longstanding concerns about how vaccines are evaluated and approved in the United States, but does not currently take a position on VLA15.
Dr. Luis Marcos, an infectious disease specialist at Stony Brook Medicine who treats many patients with Lyme and other tick-borne illnesses, said interest in the new vaccine appears strong.
“People are definitely interested in this vaccine — more than the flu, more than the COVID vaccine,” he told Newsday. “And it’s a different vaccine than the previous one.”
Top photo: (AP Photo/Gary M. Baranec)